Masood Ahmad
Tanveer Khan1*, and Kasatkin Valery2
1Faculty of Pharmacy, Jouf University, Sakaka, Kingdom of Saudi Arabia.
2Faculty of Medical Science, International University of Fundamental Studies, Moscow, Russia.
*********************
Abstract: –Excipients play a crucial role in the quality of dosage forms. Disintegrants, a widely used category of excipient, have an influence on the dissolution profile of tablets. They act mainly by two mechanisms: either swelling or capillary water absorption. Rapidly disintegrating tablets are prepared by using suitable natural superdisintegrants (SD). The researcher developed the fast disintegrating tablets (FST) by using natural SD with improved patient compliance and convenience. FST by using natural SD overcome the disadvantages of conventional dosage form especially dysphasia in pediatric and geriatric patients.This review discusses some of the most essential natural SD that are used as an excipient in drug delivery systems.
***********************
Key words:-Disintegration, Natural Superdisintegrants, Fast Dissolving Tablets
Introduction:-
Oral route is the most preferred route for administration of different drugs because it is regarded as safest, most convenient, and economical route. The tablets are the most favored oral solid dosage form In fact, it is more popular dosage form and almost 70% of medicines are dispensed in tablet form (Pahwa & Gupta, 2011) , (Vimal et al., 2014). Disintegration of dosage form are depends upon various physical factors of disintegrant/ superdisintegrant(Shihora & Panda, 2011).
Natural disintegrants constitute a group of materials that on contact with water swell, hydrate, change in volume or form or react chemically to produce a disruptive change in the tablet. Rapidly disintegrating tablets are prepared by using suitable natural superdisintegrants. Natural superdisintegrantsthe effect on the physical factor of tablet formulation. (Amaliyar et al., 2014).
The Ideal characteristic of Superdisintegrants:-
The Superdisintegrants have Poor water solubility with good hydration capacity and Poor gel formation. It also have Good compressibility, Inert, Non-toxic and Good flow properties the Superdisintegrants requirement of least quantity is that Good mouth feel and Particle size (Shihora et al., 2011).
Natural Superdisintegrants:-
The Plant products serve as an alternative to synthetic products because of local accessibility, eco-friendly nature, bio-acceptable, renewable source and lower prices compared to important synthetic products.
Majority of investigations on natural polymers for disintegrant activity are centered on polysaccharides and proteins, due to their ability to produce a wide range of materials and properties based on their molecular structures (Yadav et al., 2010).
Advantages of Natural Polymers:-
(i) Biodegradable: Biodegradable as they are naturally available, and they are produced by all living organisms.
(ii) Biocompatible and non-toxic: Basically, all of these plant materials are reiterating sugar polysaccharides
(iii) Low cost: They are cheaper to utilize as natural sources.
(iv) Environmental-friendly processing: natural compounds obtained from different plant sources which are widely utilized in pharmaceutical industry
(v) Local availability (especially in developing countries): there is promotion for the production of plants as pharmaceutical excipients being done by government, for bulk production, like gum and mucilage’s.
(vi) Patient tolerance as well as public acceptance: There is less chance with natural materials compared with synthetic one (Kumar ,S.A et al., 2012).
Natural Products utilized as Superdisintegrants.(Table#1)
Lepidium Sativum:
It is widely used as herbal medicine in India. Parts used are leaves, root, oil, seeds etc. Seeds contain higher amount of mucilage, dimeric imidazole alkaloids lepidine B, C, D, E and F and two new monomeric imidazole alkaloids semi lepidinoside A and B. Mucilage of Lepidium sativumhas various characteristic like binding, disintegrating, gelling etc. respectively (Diverkar et al., 2010).
- Fenugreek:
Commonly known as Fenugreek, is an herbaceous plant of the leguminous family and contain a high percentage of mucilage which can be used as disintegrant for use in mouth dissolving tablet formulations. Mucilage is an off white-cream yellow colored amorphous powder that quickly dissolves in warm water to form viscous colloidal solution (Kumar,R. et al., 2009).
- Guar Gum:-
The gum is commercially extracted from the seeds ,essentially by a mechanical process of roasting, differential attrition, sieving and polishing. Chemically, guar gum is a polysaccharide composed of the sugars galactose and mannose.
The backbone is a linear chain of 1, 4-linked mannose residues to which galactose residues are 1, 6-linked at every second mannose, forming short side-branches (Chaplin, 2006).
It is used as thickener, stabilizer and emulsifier and good natural superdisintegrant.It has also been investigated in the preparation of sustained release matrix tablets in the place of cellulose derivatives
- PlantagoOvata: .
Mucilage of Plantagoovatahas various characteristics like binding, disintegrating and sustaining properties. Mucilage can be used as natural superdisintegrant to formulate fast dissolving tablets because it has very high percentage of swelling index (around 89±2.2%v/v) as compared to the other super disintegrating property. (R. Kumar et al., 2009)
The separated mucilage was dried in oven at temperature less than 60°C, powdered, sieved (#80) and stored in a desiccators until use (Chakraborty et al., 2008).
- Aloe Vera:-
Aloe vera plants were separated out with the help of a sterile knife and homogenized in a blender at 30 rpm. The homogenized mass was separated with a G3 sintered glass filter under vacuum, freeze dried using a bench-top freeze-dryer and subsequently stored at 40°C. The ratio of AVG to lyophilized powder was 200:1 (Turner et al., 2004).
- Hibiscus Rosa-Sinensis:-.
The leaves were crushed and soaked in water for 5–6 h, boiled for 30 min and left to stand for 1 h to allow complete release of the mucilage into the water. The mucilage was extracted using a multi layer muslin cloth bag to remove the marc from the solution. The mucilage was separated, dried in an oven at 35°C, collected, grounded, passed through a # 80 sieve and stored in a desiccator at 30°C & 45% relative humidity till use (Wahi et al., 1985a,1985b).Natural superdisintegrants Mucilage of Hibiscus rosa-sinensis contains L‐rhamnose, D‐galactose, D‐-galactouronic acid, and D‐glucuronic acid.
- Ground Nut (Pea Nut) Shells powder:-
Shells were crushed in grinderand then passed though # 60 mesh. The Characterization of GNSP the various phytochemical tests and flow property study of only Ground Nut (Pea Nut) Shells powder was carried out [8].So we can able to use the GNSP as superdisintegrant and formulate the fast dissolving tablet in low cost and by avoiding the synthetic superdisintegrants (Harrell et al., 2010).
- Gum Karaya :-
On hydrolysis it yields galactose, rhamnose and galacturonic acid. Gum Karaya occurs as a partially acetylated derivative. It is a dried exudation of Sterculia Uren tree. Its synonyms are Karaya, sterculia, Indian tragacanth, Bassoratragacanth, kadaya, Kadira, katila. Gum Karaya is compatible with other plant hydrocolloids as well as proteins and carbohydrates and act as natural Gum.
- Cassia fistula gum:-.
Gum obtained from the seeds of Cassia fistula. Carboxymethylation as well as carbamoylethylation of Cassia gum is reported to improve cold water solubility, improve viscosity and increase microbial resistance as compared to native gum contain calcium or sodium salts of carboxymethylated or carbamoylethylatedC. fistula gum as superdisintegrant in the formulation development of FDT.
- Locust Bean gum :-
It is also called Carob bean gum. Some other familiar polysaccharides are starch and cellulose, which are made of long chains of the sugar glucose. In guar gum, giving it slightly different properties.Pharmaceutical application of locust bean gum in various novel drug delivery systems. Locust bean gum has also been reported to have bioadhesive and solubility enhancement properties. There are various reports that Locust bean gum can be used in pharmaceutical as natural superdisintegrant and biotechnological purpose.
- Mango Peel Pectin :-
. Rather mango peel pectin cannot be used for promising the behavior of superdisintegrants, but due to its good swelling index and good solubility in biological fluids it can be used to prepare fast dispersible tablets (Shihora & Panda, 2011),(Mangal et al., 2012).
- Asaliya:-
Pharmaceutical field as disintegrating agent and as herbal medicine. Seeds contain a major proportion of mucilage, the mucilage can be extracted from seeds by different procedures and its yield varies from 14% to 22%. Mucilage characteristic like binding, disintegrating, gelling etc. The extracted mucilage is used to develop fast dissolving tablets.
The values of swelling index, angle of repose, bulk density and tapped density are estimated as following 18, 32ºC, 0.58g/cc and 0.69g/cc respectively (Deshmkh et al., 2012).
- Gellan Gum:-
It is a linear anionic polysaccharide biodegradable polymer. Gellan gum act as a superdisintegrant and the efficiency of gellan gum is batter as compared with other conventional disintegrants. The disintegration of tablet might be due to the instantaneous swelling characteristics of gellan gum when it comes into contact with water and owing to its high hydrophilic nature.
The complete disintegration of tablet is observed within 4 minutes with gellan gum has been proved itself as a superdisintegrant (Zhang et al., 2010).
- Alginates:-
These are hydrophilic colloidal substances extracted naturally from certain species of Kelp or chemically modified from natural sources like alginic acid or salt of alginic acid. They are having higher affinity for water absorption and capable for an excellent disintegrants. They can be successfully used in ascorbic acid, multivitamins formulation as Superdisintegrant.
- Modified Polysaccharides:-
Agar (AG) and guar gum (GG), natural polysaccharides are treated with water and co grinded further with mannitol which exhibit superdisintegration property. These modified polysaccharides may call C-TAG (co grinded treated agar) and C-TGG (co grinded treated guar gum) respectively.(Shihora & Panda, 2011), (Sharma et al., 2002).
- Agar and Treated Agar:-
It is the dried gelatinous substance obtained from Gelidiumamansii(Gelidanceae) and several other species of red algae like Gracilaria(Gracilariaceae) and Pterocladia (Gelidaceae). Agar consists of two polysaccharides, agarose and agar pectin. Agarose is responsible for gel vigor and agar pectin is responsible for the viscosity of agar solutions. High gel vigor of agar makes it a potential candidate as a disintegrants (Setia et al., 2011)
- FicusIndica Fruit Mucilage:-
Themucilage of ficusindica fruit is utilized as superdisintegrant which is obtained from the pulp of fruit ficusindica.
It has nutritional as well as medicinal value. The dried and uncooked ficusindica fruit gives 230 kcal (963 KJ) of energy per 100 gm or 3.5 oz. (ounce). It is utilized in assuaging fever, pain, inflammation, wound rejuvenating, blood quandaries, and urinary quandaries.
- Mangiferaindica Gum :-
Mundane name of Mangiferaindicais mango, and it belongs to Anacardiaceae family.It is nontoxic and utilized as disintegrant, binder, suspendingagent, and emulsifying agent in different formulations. The gum powder is white to off white in color, and the powder was soluble in water and virtually insoluble in acetone chloroform, ether, methanol, and ethanol. It is facilely available, and gum is devoid of toxicity, and each and every component of the tree has pharmacological activity like diuretic, astringent, diabetes, asthma, diarrhea, urethritis, and scabies (Alam et al., 2012)
- Dehydrated Banana Powder :-
Banana is additionally called plantain. DBP is yare from the variety of banana called Ethan and nenthran (nenthravazha) and belongs to the family Musaceae. It contains vitamin A, so it is utilized in the treatment of gastric ulcer and diarrhea. It withal contains vitamin B6, which avails in reducing the stress and solicitousness. It is a very good source of energy due to high carbohydrate content, and it contains potassium, which is responsible for more preponderant brain functioning(Arun, 2013).
- Soy polysaccharide:-
It is a natural superdisintegrant that does not contain any starch or sugar so can be used in nutritional products. (Iqubal et al., 2014). Evaluated soy polysaccharide (a group of high molecular weight polysaccharides obtained from soy beans) as a disintegrant in tablets made by direct compression using lactose and dicalcium phosphate dihydrate as fillers. A cross-linked sodium carboxy-methyl cellulose and corn starch were used as control disintegrants. Soy polysaccharide performs well as a disintegrating agent in direct compression formulations with results paralleling those of cross-linked CMC.(Younes & Rinaudo, 2015),(Kumar,T. et al., 2012)
- Chitin and Chitosan :-
Chitosan is produced commercially by deacetylation of chitin, which is the structural element in the exoskeleton of crustaceans (such as crabs and shrimp) and cell walls of fungi. (Bruscato et al 1978) reported that when chitin was included in the conventional tablets, the tablets disintegrated with in 5 and 10 minutes irrespective of solubility of the drug. The disintegration time in the oral cavity as well as wetting time could be analyzed by surface free energy. Chitosan is the best known natural polysaccharide used for its versatile applications as natural superdisintegrant in pharmaceutical industry(Iqubal et al., 2014).
- Pectin:-
Pectin is a family of galacturonic acid-rich polysaccharides including homogalacturonan .Pectin is structurally and functionally the most complex polysaccharide in plant cell walls. Pectin has functions in plant growth, morphology, development, and plant defense and also serves as a gelling and stabilizing polymer in diverse food and specialty products and has positive effects on human health and multiple biomedical uses. (Mohnen, 2008).
Co-processed Superdisintegrant:-
The New and improved superdisintegrants continue to be developed to meet the needs of advanced tablet manufacturing. It requires the development of various added functionality excipients, which are used to achieve formulations with desired end effects. Until now only superdisintegrants are available to prepare the dosage forms, but now days different blend of excipients are available which can give disintegration property. Someco-processed excipients blends are designed to satisfy the need of more than one excipient.
Co-processed blends of excipients:-
It involves the mixture blend of more than two excipients to satisfy the required quality using different technique like spray drying and freeze drying.
Modified Chitosan with silicon dioxide:-
This is the new excipients based on co-precipitation of Chitosan and silica. The physical interaction between Chitosan and silica create an insoluble, hydrophilic highly absorbent material, resulting in superiority in water uptake, water saturation for gelling formation. Studies have shown that Chitosan–silica delivers superior performance in wet granulation formulations and is the only disintegrant that is effective at all concentrations in tablet formulation.
Modified sugars:-
Glucidex IT:-
Glucidex IT is obtained by moderate hydrolysis of starch. It is micro granulated form enables almost instantaneous dispersal and dissolution in water. Different range of Glucidex IT products is available.
All co-processed and modified excipients are playing a vital role in the development of easy dosage forms which are resistant to atmosphere. The improved physical, chemical and mechanical properties of such excipients as compared to existing excipients, have helped in solving formulation problems such as flowability, compressibility, hygroscopicity, palatability, dissolution, disintegration, sticking, and dust generation(Pahwa & Gupta, 2011).
A brief detail about the source and graphical representation of natural superdisintegrants is presented in Table#1,2 and Fig #1.Respectively.
Table#1 Source OfNatural Superdisintegrants:-
| S.No. | Name:- | Description (Sources):- |
| 1 | Agar and Treated Agar. | Gelidiumamansii (Gelidanceae). |
| 2 | Alginates. | Extracted naturally from species of Kelp. |
| 3 | Aloe Vera. | The inner mucilaginous parenchymatous tissues of leaves of Aloe Vera. |
| 4 | Asaliya. | Seeds contain a major proportion of mucilage. (FamilyCruciferae). |
| 5 | Cassia fistula gum. | Seeds of Cassia fistula gum. |
| 6 | Chitin and chitosan . | Crab and Shrimp shells. |
| 7 | Dehydrated Banana Powder . | Ethan and nenthran(family Musaceae). |
| 8 | Fenugreek. | Trigonellafoenum-graceum (family Leguminosae). |
| 9 | FicusIndica Fruit Mucilage. | The mucilage of ficusindica fruit. |
| 10 | Gellan Gum. | Gellan gum is obtained from Pseudomonos elodea. |
| 11 | Ground Nut (Pea Nut). | Ground Nuts – Shells powder. |
| 12 | Guar Gum. | Guar gum comes from the endosperm of the seed of the legume plant |
| 13 | Gum Karaya. | Gum Karaya is a negative colloid and a complex polysaccharide |
| 14 | Hibiscus rosa-sinensis. | The fresh Hibiscus rosasinesis leaves (Family Malvaceae). |
| 15 | LepidiumSativum. | Lepidiumsativum (family: Cruciferae) Seeds |
| 16 | Locust Bean gum. | Locust bean gum is extracted from the endosperm of the seeds |
| 17 | Mangiferaindica Gum. | Mangiferaindicais mango(Family Anacardiaceae.) |
| 18 | Mango Peel Pectin. | Dried mango peel powder is use for extracting pectin |
| 19 | Modified Polysaccharides. | Natural polysaccharides. |
| 20 | PlantagoOvata. | Husk consists of dried seeds of the plantplantago ovate. |
| 21 | Pectin. | Polysaccharide in plant cell walls. |
| 22 | Soy polysaccharide. | Soy beans |
Examples of chemical structure for some of these natural superdisintegrants are represented inTable#2
Table#2 Chemical Structure Of Natural Superdisintegrants.
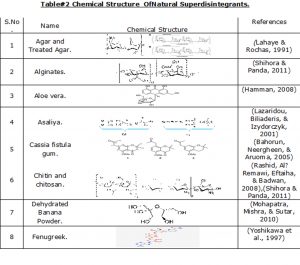
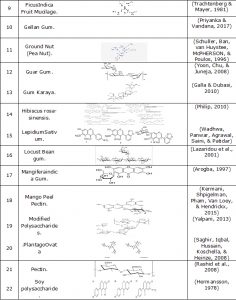
| S.No. | Name | Chemical Structure | References |
| 1 | Agar and Treated Agar. | (Lahaye & Rochas, 1991) | |
| 2 | Alginates. | (Shihora & Panda, 2011) | |
| 3 | Aloe vera. | (Hamman, 2008) | |
| 4 | Asaliya. | (Lazaridou, Biliaderis, & Izydorczyk, 2001) | |
| 5 | Cassia fistula gum. | (Bahorun, Neergheen, & Aruoma, 2005) | |
| 6 | Chitin and chitosan. | (Rashid, Al‐Remawi, Eftaiha, & Badwan, 2008),(Shihora & Panda, 2011) | |
| 7 | Dehydrated Banana Powder. | (Mohapatra, Mishra, & Sutar, 2010) | |
| 8 | Fenugreek. | (Yoshikawa et al., 1997) | |
| 9 | FicusIndica Fruit Mucilage. | (Trachtenberg & Mayer, 1981) | |
| 10 | Gellan Gum. | (Priyanka & Vandana, 2017) | |
| 11 | Ground Nut (Pea Nut). | (Schuller, Ban, van Huystee, McPHERSON, & Poulos, 1996) | |
| 12 | Guar Gum. | (Yoon, Chu, & Juneja, 2008) | |
| 13 | Gum Karaya. | (Galla & Dubasi, 2010) | |
| 14 | Hibiscus rosa-sinensis. | (Philip, 2010) | |
| 15 | LepidiumSativum. | (Wadhwa, Panwar, Agrawal, Saini, & Patidar) | |
| 16 | Locust Bean gum. | (Lazaridou et al., 2001) | |
| 17 | Mangiferaindica Gum. | (Arogba, 1997) | |
| 18 | Mango Peel Pectin. | (Kermani, Shpigelman, Pham, Van Loey, & Hendrickx, 2015) | |
| 19 | Modified Polysaccharides. | (Yalpani, 2013) | |
| 20 | .PlantagoOvata | (Saghir, Iqbal, Hussain, Koschella, & Heinze, 2008) | |
| 21 | Pectin. | (Rashid et al., 2008) | |
| 22 | Soy polysaccharide. | (Hermansson, 1978) |
Fig#1Graphical Representation of Source Of Natural Superdisintegrants
Others Material use as Super disintegrating agents,-
Super disintegrating agents, which provides superior disintegration, the research for newer disintegrating agents is continuing and researchers are testing with modified natural products, similar to formalin casein, chitin, chitosan, polymerized agar acrylamide, xylem, cross-linked carboxymethylguar and modified tapioca starch. Studies have proposed that the water insoluble superdisintegrating agents show improved disintegration property than the to some extent water soluble agents, since they do not have an affinity to swell. Superdisintegrating agents that have a tendency to swell show minor retardation of the disintegration property due to formation of sticky barrier. There is no certain upper limit regarding the amount of superdisintegrating agent as long as the mechanical properties of the tablet are well-matched with its intended use. The super disintegrating agent may be used alone or in combination with other superdisintegrating agent. (Nasir et al., 2017)
Conclusion:-
All of natural superdisintegrants are available in the market at a very low price and we easily use in novel product. Orally disintegrating tablets which are formulated by the use of natural superdisintegrants to develop tablet formulations having rapid disintegration, pleasant mouth feel and high breaking force for tablet robustness. Have a great appeal to consumers and increased compliance among users. Fast Dissolving tablets disintegration of dosage forms depends upon various physical factors of natural superdisintegrants percentage used, proportion of disintegrants used, compatibility with other excipients and presence of surfactants and hardness of the tablets, nature of drug substances and mixing and types of addition. These all factor covered by the use of natural superdisintegrantseasy to isolate and economically to be used in tablet formulation.
References:-
Alam, M. T., Parvez, N., & Sharma, P. K. (2014). FDA-Approved natural polymers for fast dissolving tablets. Journal of pharmaceutics,Article ID #952970.
Amaliyar, P. R., Patel, H., Chaudhary, S. A., Shah, H., Patel, A., & Suva, M. A. (2014). A brief review on natural and synthetic superdisintegrants. Inventi Rapid: Novel Excipients, 3, 1-6.
Arogba, S. S. (1997). Physical, chemical and functional properties of Nigerian mango (Mangifera indica) kernel and its processed flour. Journal of the Science of Food and Agriculture, 73(3), 321-328.
Arun, R. (2013). Comparative evaluation of potato starch and banana powder as disintegrating agents in Aceclofenac tablet formulation. International Journal of Pharmaceutical Science, 5(2), 204-207.
Bahorun, T., Neergheen, V. S., & Aruoma, O. I. (2005). Phytochemical constituents of Cassia fistula. African journal of Biotechnology, 4(13).
Chakraborty, S., Khandai, M., Singh, S. P., & Patra, N. C. (2008). Comparative study on effect of natural and synthetic superdisintegrants in the formulation of fast dissolving tablets. International Journal of Green Pharmacy (IJGP), 2(1).
Chaplin, M. (2006). Water structure and behavior: guar gum. London: South Bank University.
Deshmkh, H., Chandrashekhara, S., Nagesh, C., Murade, A., & Usgaunkar, S. (2012). Superdisintegrants: A recent investigation and current approach. Asian Journal of Pharmacy and Technology, 2(1), 19-25.
Divekar Varsha, B., & Mohan, K. (2010). Isolation and characterization of mucilage from lepidium sativum linnseeds. Internaional journal of pharma research and development, 2, 1-5.
Galla, N. R., & Dubasi, G. R. (2010). Chemical and functional characterization of Gum karaya (Sterculia urens L.) seed meal. Food Hydrocolloids, 24(5), 479-485.
Hamman, J. H. (2008). Composition and applications of Aloe vera leaf gel. Molecules, 13(8), 1599-1616.
Harrell, T., Diaz, N., Jayanathan, S., Sevilla, S., & Burwen, J. (2010). Developing Peanut Shell Fuel Briquettes for Household Use in Malawi. CE290: Design for Sustainable Development, 2-39.
Hermansson, A.-M. (1978). Physico‐chemical aspects of soy proteins structure formation. Journal of Texture Studies, 9(1‐2), 33-58.
Iqubal, M. K., Singk, P., Shuaib, M., & Singh, M. (2014). Recent advances in direct compression technique for Pharmaceutical tablet formulation. International journal of pharmaceutical research and development, 6(1), 49-57.
Kermani, Z. J., Shpigelman, A., Pham, H. T. T., Van Loey, A. M., & Hendrickx, M. E. (2015). Functional properties of citric acid extracted mango peel pectin as related to its chemical structure. Food Hydrocolloids, 44, 424-434.
Kumar, R., Patil, S., Patil, M., Patil, S. R., & Paschapur, M. S. (2009). Isolation and evaluation of disintegrant properties of fenugreek seed mucilage. International Journal of PharmTech Research, 1(4), 982-996.
Kumar, S. A., Vivek, D., & Vandana, A. (2012). Role of natural polymers used in floating drug delivery system. Journal of Pharmaceutical and Scientific Innovation, 1(3), 11-15.
Kumar, T., Gupta, S. K., Prajapati, M. K., & Tripathi, D. (2012). Natural excipients: A review. Asian Journal of Pharmacy and Life Science ISSN, 2231, 4423.
Lahaye, M., & Rochas, C. (1991). Chemical structure and physico-chemical properties of agar. Paper presented at the International workshop on gelidium.
Lazaridou, A., Biliaderis, C. G., & Izydorczyk, M. S. (2001). Structural characteristics and rheological properties of locust bean galactomannans: a comparison of samples from different carob tree populations. Journal of the Science of Food and Agriculture, 81(1), 68-75.
Mangal, M., Thakral, S., Goswami, M., & Ghai, P. (2012). Superdisintegrants: An updated review. International journal of pharmacy and pharmaceutical science research, 2(2), 26-35.
Mohapatra, D., Mishra, S., & Sutar, N. (2010). Banana and its by-product utilisation: an overview.
Mohnen, D. (2008). Pectin structure and biosynthesis. Current opinion in plant biology, 11(3), 266-277.
Nasir, A., Gohar, U. F., & Ahmad, B. A Review Article on Superdisintegrants.
Pahwa, R., & Gupta, N. (2011). Superdisintegrants in the development of orally disintegrating tablets: a review. International journal of pharmaceutical sciences and research, 2(11), 2767.
Philip, D. (2010). Green synthesis of gold and silver nanoparticles using Hibiscus rosa sinensis. Physica E: Low-Dimensional Systems and Nanostructures, 42(5), 1417-1424.
Priyanka, S., & Vandana, S. (2017). A review article on: superdisintegrants. International Journal of Drug Research and Technology, 3(4), 11.
Rashid, I., Al‐Remawi, M., Eftaiha, A., & Badwan, A. (2008). Chitin–silicon dioxide coprecipitate as a novel superdisintegrant. Journal of Pharmaceutical Sciences, 97(11), 4955-4969.
Saghir, S., Iqbal, M. S., Hussain, M. A., Koschella, A., & Heinze, T. (2008). Structure characterization and carboxymethylation of arabinoxylan isolated from Ispaghula (Plantago ovata) seed husk. Carbohydrate polymers, 74(2), 309-317.
Saini, P., & Sharma, N. (2012). 3. Natural Polymers used in Fast Disintegrating Tablets: A Review. International Journal of Drug Development and Research, 4(4).
Schuller, D. J., Ban, N., van Huystee, R. B., McPHERSON, A., & Poulos, T. L. (1996). The crystal structure of peanut peroxidase. Structure, 4(3), 311-321.
Setia, A., Goyal, N., & Kansal, S. (2011). Formulation and evaluation of Ciprofloxacin hydrochloride dispersible tablets using natural substances as disintegrates. Pelagia Research Library Der Pharmacia Sinica, 2(1), 36-39.
Sharma, V., Philip, A. K., & Pathak, K. (2008). Modified polysaccharides as fast disintegrating excipients for orodispersible tablets of roxithromycin. AAPS PharmSciTech, 9(1), 87.
Shihora, H., & Panda, S. (2011). Superdisintegrants, utility in dosage forms: a quick review. J Pharm Sci Biosci Res, 1, 148-153.
Shu, T., Suzuki, H., Hironaka, K., & Ito, K. (2002). Studies of rapidly disintegrating tablets in the oral cavity using co-ground mixtures of mannitol with crospovidone. Chemical and pharmaceutical bulletin, 50(2), 193-198.
Trachtenberg, S., & Mayer, A. M. (1981). Composition and properties of Opuntia ficus-indica mucilage. Phytochemistry, 20(12), 2665-2668.
Turner, C. E., Williamson, D. A., Stroud, P. A., & Talley, D. J. (2004). Evaluation and comparison of commercially available Aloe vera L. products using size exclusion chromatography with refractive index and multi-angle laser light scattering detection. International immunopharmacology, 4(14), 1727-1737.
Vimal, V., Aarathi, T., & Anuja, S. B. (2013). Superdisintegrants in fast disintegrating drug delivery systems: A brief review. Int. J. Pharm, 3(2), 380-385.
Wadhwa, S., Panwar, M., Agrawal, A., Saini, N., & Patidar, L. P. L. A review on pharmacognostical study of lepidium sativum lepidium sativum.
Wahi, S., Sharma, V., Jain, V., & Sinha, P. (1985a). Studies on emulsifying property of mucilages of Hygrophila spinosa and Hibiscus esculentus. Indian J. Natural Product, 1, 3-6.
Wahi, S., Sharma, V., Jain, V., & Sinha, P. (1985b). Studies on suspending property of mucilage of Hygrophila Spinosa T. Anders and Hibiscus Esculents Linn. Indian Drug, 22, 500-502.
Yadav, N. D., Pingale, P. L., & Tatane, S. R. (2010). Comparative study on effect of natural and artificial superdisintegrants in the formulation of fast dissolving aspirin tablet. Journal of Pharmacy Research, 3(7), 1594-1597.
Yalpani, M. (2013). Polysaccharides: syntheses, modifications and structure/property relations (Vol. 36): Elsevier.
Yoon, S.-J., Chu, D.-C., & Juneja, L. R. (2008). Chemical and physical properties, safety and application of partially hydrolized guar gum as dietary fiber. Journal of Clinical Biochemistry and Nutrition, 42(1), 1-7.
Yoshikawa, M., Murakami, T., Komatsu, H., murakami, n., yamahara, j., & matsuda, h. (1997). Medicinal foodstuffs. IV. Fenugreek seed.(1): structures of trigoneosides Ia, Ib, IIa, IIb, IIIa, and IIIb, new furostanol saponins from the seeds of Indian Trigonella foenum-graecum L. Chemical and pharmaceutical bulletin, 45(1), 81-87.
Younes, I., & Rinaudo, M. (2015). Chitin and chitosan preparation from marine sources. Structure, properties and applications. Marine drugs, 13(3), 1133-1174.
Zhang, Y., Wrzesinski, A., Moses, M., & Bertrand, H. (2010). Comparison of superdisintegrants in orally disintegrating tablets. Pharmaceutical Technology, 34(7), 54-65.